Vancomycin is an antibiotic used to treat serious bacterial infections. It’s most commonly administered intravenously, typically for infections such as bacteremia, endocarditis, osteomyelitis, etc. Despite its wide-spread use and success in treating these serious infections, clinicians have a challenging job to provide the correct dosage to their patients. This is because the manner in which Vancomycin distributes through the body is complex, with factors such as body weight, muscle mass, and fat distribution all playing a role. Vancomycin also has a narrow therapeutic index. This means that small differences in dose or blood concentration can lead to therapeutic failures or adverse drug reactions and an increased risk of overdosing or underdosing.
Vancomycin dosing guidelines therefore recommend close adjustments based on total-body-weight. However recent research shows that this is not ideal for obese patients. While the volume of distribution of vancomycin increases with body weight, it does not increase proportionally. The results of incorrect dosing of Vancomycin are serious. Overdosing can lead to liver toxicity, hearing loss, and blood clots, for example, and underdosing to prolonged hospitalization, even treatment failure.
A new approach to dosing Vancomycin for obese patient populations using so-called AUC-guided monitoring method produces more accurate dosing. However, these calculations are often time consuming and can be a major hindrance in scenarios where time to antibiotic administration is critical. Due to the recent nature of this newly recommended dosing approach, there is a lack of automated tools available for use in clinical practice at this time.
In this article I look at how I was able to overcome the challenges of calculating Vancomycin dosing for patients with obesity and build an automated Vancomycin dosage calculator. The calculator incorporates the customized AUC-guided requirements, gives clinicians access to precise dosage recommendations, and the ability to respond quickly to insight from Therapeutic Drug Monitoring (TDM).
The Importance of Therapeutic Drug Monitoring with Vancomycin
Vancomycin’s narrow therapeutic index requires that Vancomycin dosing is monitored according to Therapeutic Drug Monitoring (TDM) and dosings adjusted quickly and accurately.
TDM allows clinicians to measure and track drug levels in the patient’s body throughout the course of treatment. By tracking these levels, clinicians are better able to dose Vancomycin within its therapeutic index, and reduce the risks of over- or underdosing.
This monitoring of drug levels involves the collection of blood samples at specified times during treatment, followed by laboratory testing, and the use of pharmacokinetic-pharmacodynamic (PKPD) calculations to determine the true Vancomycin concentration in the blood – what the drug is currently “doing” to the body.
Area-Under-the-Curve (AUC) Monitoring
The most common PKPD parameters used to predict the concentration of Vancomycin in the blood include the Minimum Inhibitory Concentration (MIC), peak and trough levels, and the area-under-the-curve (AUC).
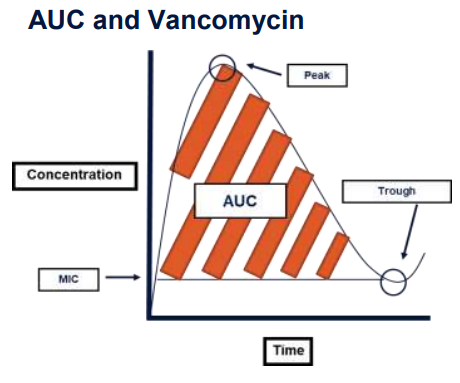
The MIC is the lowest concentration of antibiotic which prevents visible growth of bacteria. This concentration is dependent on the patient, the antibiotic, and the offending bacterial pathogen, which may vary across different clinical scenarios. The MIC for vancomycin is assumed to be 1 mg/L in most clinical scenarios.
The peak level is the highest concentration of antibiotic reached after administration, whereas the trough level is the lowest concentration of antibiotic reached.
The AUC refers to the total exposure of antibiotic over a specified period of time, also known as the dosing interval. Certain antibiotics, such as vancomycin, are considered to be time dependent, and demonstrate the best bacterial inhibition when the total antibiotic exposure remains above the target MIC.
Therefore, the best predictor of Vancomycin activity in the body is an AUC/MIC ratio and can be used to guide dosing decisions.
Challenges in Calculating Vancomycin
Previous vancomycin guidelines recommended using trough-guided monitoring of vancomycin, in which trough levels were used as a surrogate measure of the patient’s AUC (2). This method suggested that a trough level of >10 mg/L would be sufficient to sustain an AUC goal of >400 mg*h/L, which is the specific AUC at which vancomycin is determined to be effective.
However, further research has determined that trough levels have poor correlation with the AUC, often resulting in underestimation and increased antibiotic exposure.
Recent updates to the vancomycin guidelines place a strong emphasis on the use of AUC-guided monitoring in patients with severe MRSA infections, who will be receiving >5 days of therapy, and may be at increased risk of liver toxicity (3). This monitoring method involves the collection of peak and trough levels, followed by laboratory testing, and PKPD calculations of the patient’s true AUC. For these patients, an AUC of 400-600 mg*h/L should be targeted.
New Approach to Vancomycin Calculation Helps Obese Patient Populations
Recent research from Masich et. al.(4), further expands upon this recommendation as it relates to the patient population with obesity, where certain PKPD parameters may be altered from the standard patient population. Studies indicate that while the volume of distribution of vancomycin does increase with body weight, it does not increase proportionally.
In patients with obesity, the distribution of vancomycin in the body may increase or decrease depending on patient specific factors, such as body fat content, muscle mass, and kidney function. In her research, Masich develops a novel approach to dosing vancomycin in this patient population using the newly emphasized AUC-guided monitoring method.
However, these calculations can often be time consuming in the clinical setting and can be a hindrance in scenarios where time to antibiotic administration is critical. The transition to AUC-guided monitoring adds steps and calculations that may not have been previously relied upon. Additionally, due to the recent nature of this recommendation, there is a lack of automated tools available for use in clinical practice at this time. Standard IT approaches to automating this type of calculation are simply not suited to the individualized nature of Vancomycin dosing for patients with obesity.
The use of an automated tool for vancomycin calculations would overcome several barriers in clinical practice: time constraints, dosing inaccuracies, and special population considerations.
How a Customized, Automated Tool Enables Precise Treatment
The complex nature of vancomycin dosing often requires a reliance upon healthcare professionals with an in-depth knowledge of PKPD parameters. A code free data analytics platform, such as KNIME Analytics Platform (5), can be used to build a workflow that makes the calculations with the necessary customization. Advanced data science tools provide the flexibility to build such a customized and complex application. KNIME’s visual programming environment bridges the coding skills gap. Healthcare professionals can build analytics applications without needing to know how to code and are able to fold their valuable expertise into the process.
The Vancomycin Calculator in KNIME
Over the span of a five-week advanced pharmacy practice experience (APPE) in digital health, a PharmD candidate with no prior knowledge of data analytics or coding, has successfully created an interactive vancomycin AUC-based dosing calculator for use in patients with obesity.
The underlying workflow comprises a series of components, which encapsulate the different calculation routes necessary for the patient’s final AUC estimation.
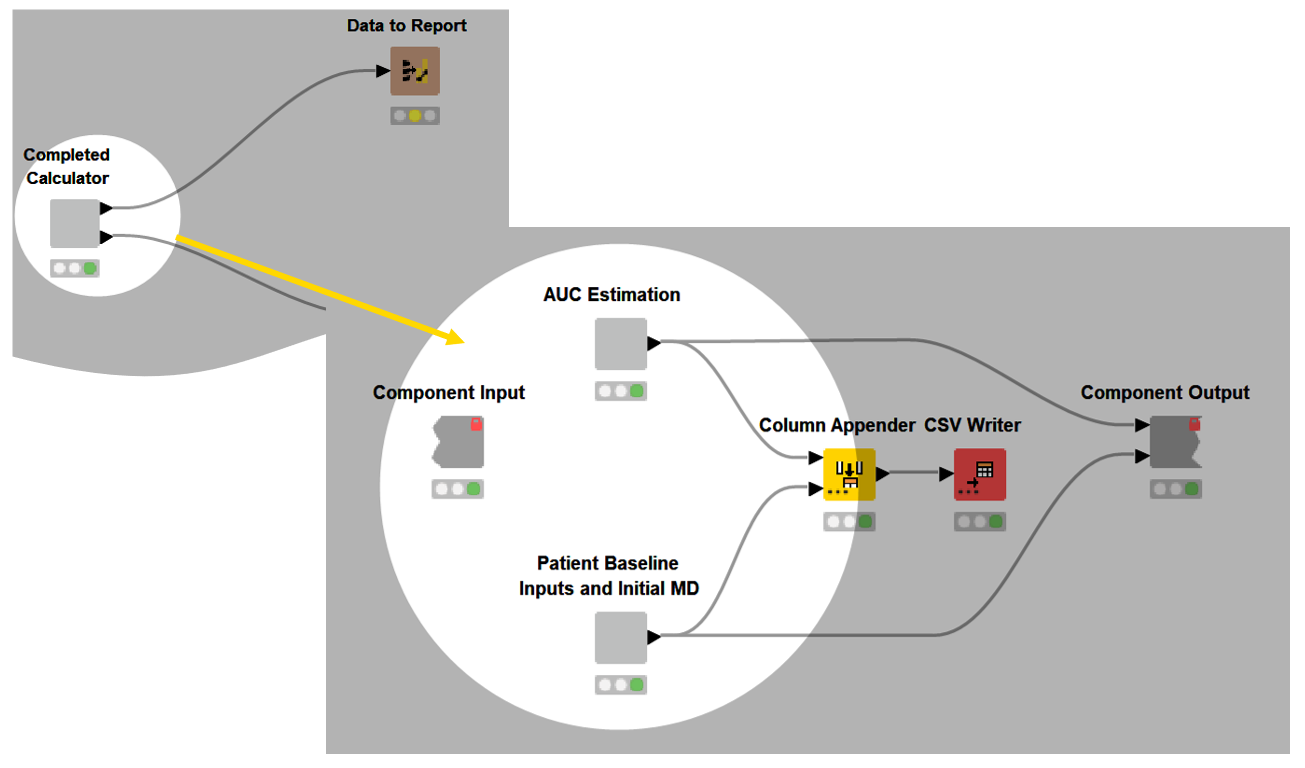
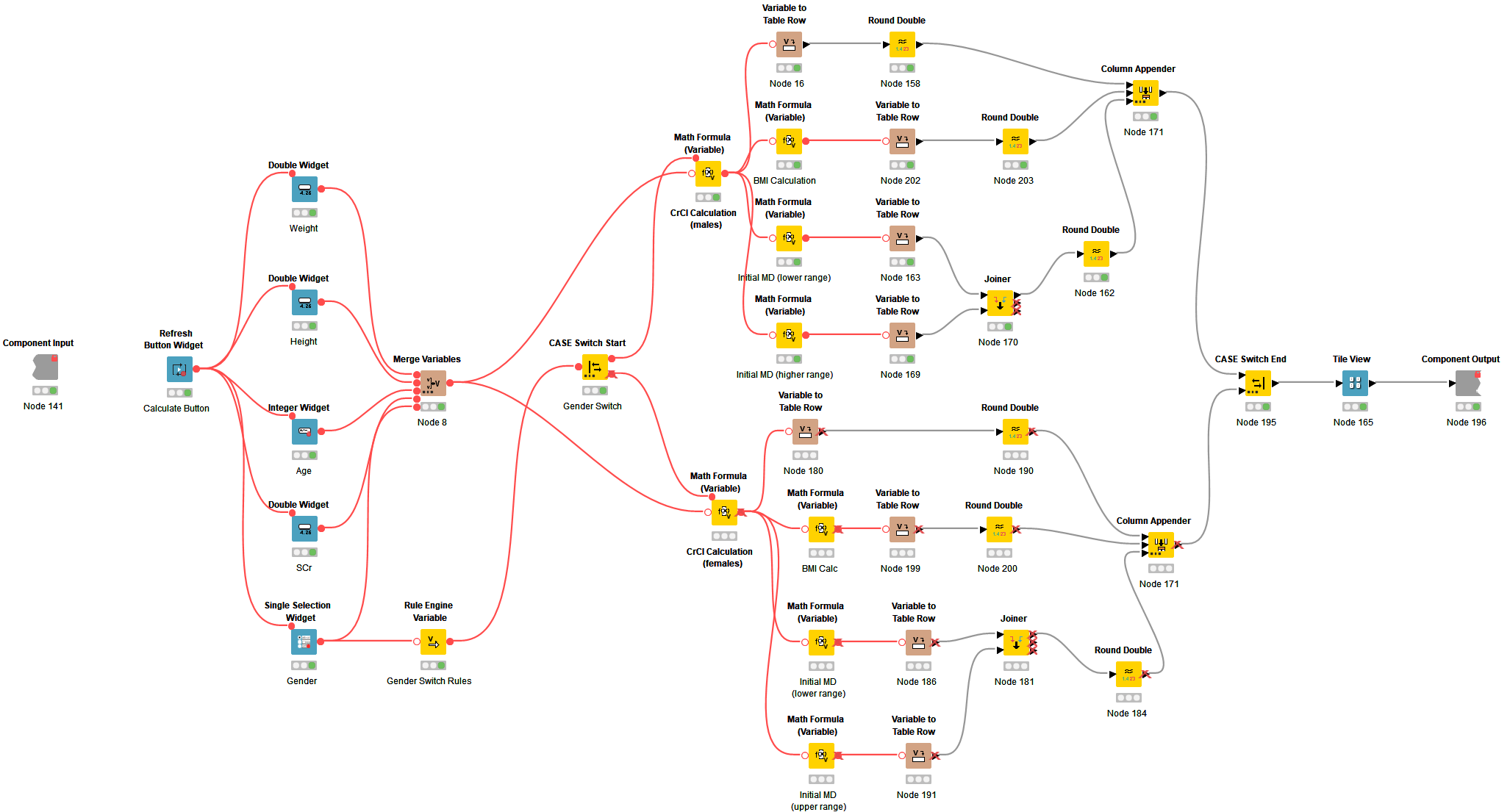
Moving through the workflow, you’ll see a component that collects a patient’s baseline inputs and calculates an initial maintenance dose (MD) of vancomycin. This component also allows for the calculation of other individualized data, such as body mass index (BMI) and creatinine clearance (CrCl). The widget nodes and tile view node included here begin to build the interactive dashboard.
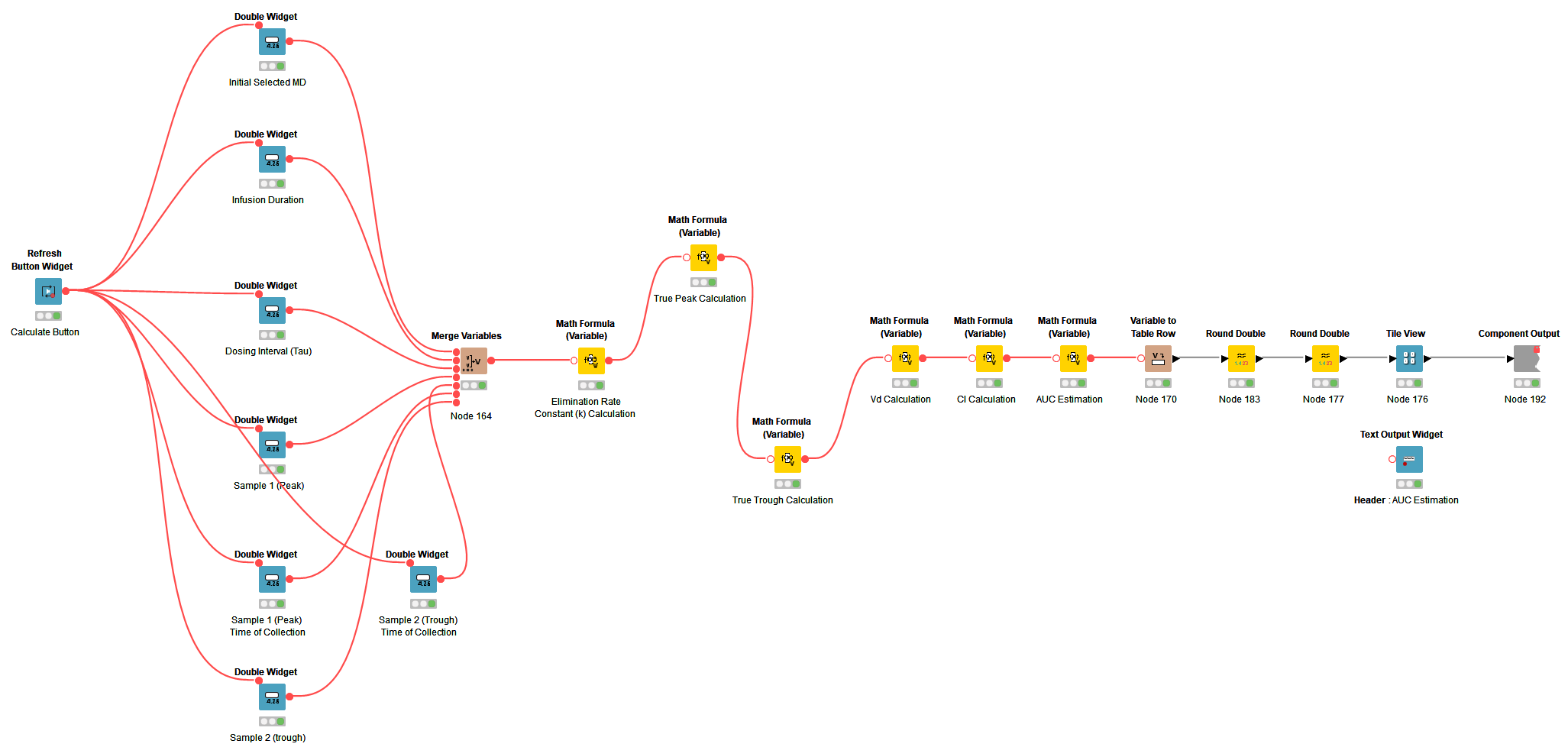
The next component of the workflow handles the first order PKPD calculations. This component encapsulates many different equations essential for estimating the biggest output of interest, which is the patient’s AUC concentration. In a similar fashion, the widget nodes and tile view nodes add functionality to the interactive dashboard.
Each of these components serve as the building blocks for the interactive calculator dashboard, which provides the functionality to individualize dosing for each patient.
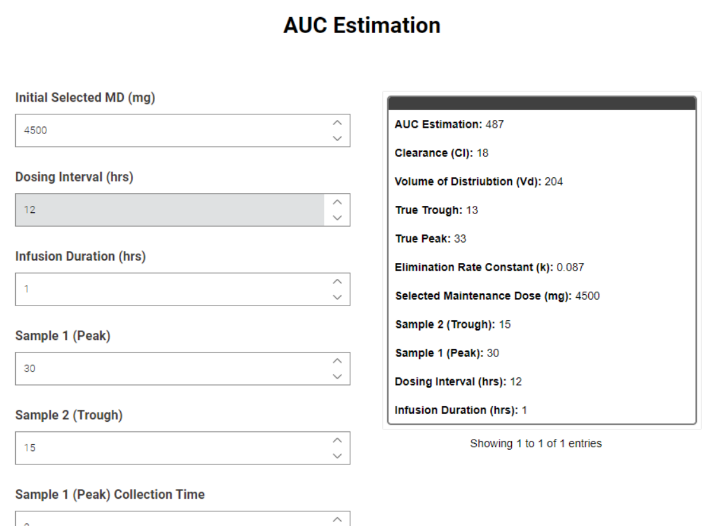

AUC-based Dosing Calculation Reduced to Seconds
The developed KNIME workflow captures each PKPD calculation step used in the estimation of a patient’s AUC, and appropriately outputs values which can be used to guide clinical decision making. These calculations, which are usually time consuming, are condensed to seconds. This allows clinicians to visualize different data inputs in a more efficient and agile manner, simplifying the vancomycin dosing process in clinical practice.
Agile Vancomycin Dosing for Better Patient Outcomes
By optimizing dosing based on the calculator, we are going to be able to prevent toxicities but also prevent the overtreatment that can happen if we don't reach the therapeutic levels. Ultimately this will result in improved patient outcomes, decreased hospitalization length, and increased time to recovery.
Special thanks to Alexander Smart PharmD, BCIDP and Anne Masich PharmD, BCPS for their support and feedback throughout.
References:
- Adult Vancomycin AUC-Guided Dosing Playbook. HCA Healthcare. Created March 1, 2022. Accessed July 13, 2022.
- Rybak MJ et al. Vancomycin Therapeutic Guidelines: A Summary of Consensus Recommendations from the Infectious Diseases Society of America, the American Society of Health-System Pharmacists, and the Society of Infectious Diseases Pharmacists. Clinical Infectious Diseases. 2009; 49(3): 325–327. https://doi.org/10.1086/600877.
- Rybak MJ et al. Therapeutic Monitoring of Vancomycin for Serious Methicillin-resistant Staphylococcus aureus Infections: A Revised Consensus Guideline and Review by the American Society of Health-system Pharmacists, the Infectious Diseases Society of America, the Pediatric Infectious Diseases Society, and the Society of Infectious Diseases Pharmacists. Clinical Infectious Diseases. 2020; 71(6): 1361–1364. https://doi.org/10.1093/cid/ciaa303.
- Masich AM et al. Vancomycin Pharmacokinetics in Obese Patients with Sepsis or Septic Shock. Pharmacotherapy. 2020; 40(3): 211-220. doi:10.1002/phar.2367.
- Berthold, M. R. et al. KNIME: The Konstanz Information Miner. Data Analysis, Machine Learning and Applications: Proceedings of the 31st Annual Conference of the gesellschaft für Klassifikation E.V., Albert-Ludwigs-Universität Freiburg. 2007; 319–326. https://doi.org/10.1007/978-3-540-78246-9_38
