Pharmacokinetics is a branch of pharmacology that provides insight on how the body responds to a drug. It encompasses processes such as Absorption, Distribution, Metabolism, and Excretion (ADME), which can be used to determine how efficient and how safe a drug is. Clinicians use pharmacokinetic parameters and calculations to visualize and interpret each of the phases of ADME as they monitor a drug’s action in vivo.
Each phase of ADME is associated with specific pharmacokinetic parameters. For example, the absorption and distribution phases of ADME can be extrapolated from drug concentrations and volumes, while the metabolism and excretion phases may be illustrated by parameters such as the elimination rate constant and clearance. Other considerations include the route and method of drug administration, which also plays a role in the body’s response to a drug. In practice, each of these pharmacokinetic parameters can be calculated, and a treatment approach can be individualized for patients.
However, one of the challenges in pharmacokinetics (PK) is the time and computational power these calculations require.
Using an automated tool in clinical practice can provide a more efficient process of drug monitoring via pharmacokinetic parameters. I used the code free data analytics platform, KNIME, to create a customized workflow capable of performing these pharmacokinetic calculations. KNIME is a platform which provides users, who may have minimum knowledge of coding and analytics, with the necessary tools to build such a calculator.
A KNIME Workflow for Pharmacokinetic Calculation
Pharmacokinetic calculation differs depending on how the drug is administered. My pharmacokinetic workflow consists of a single component, which contains different calculations routes of calculations, necessary for estimating different pharmacokinetic parameters.

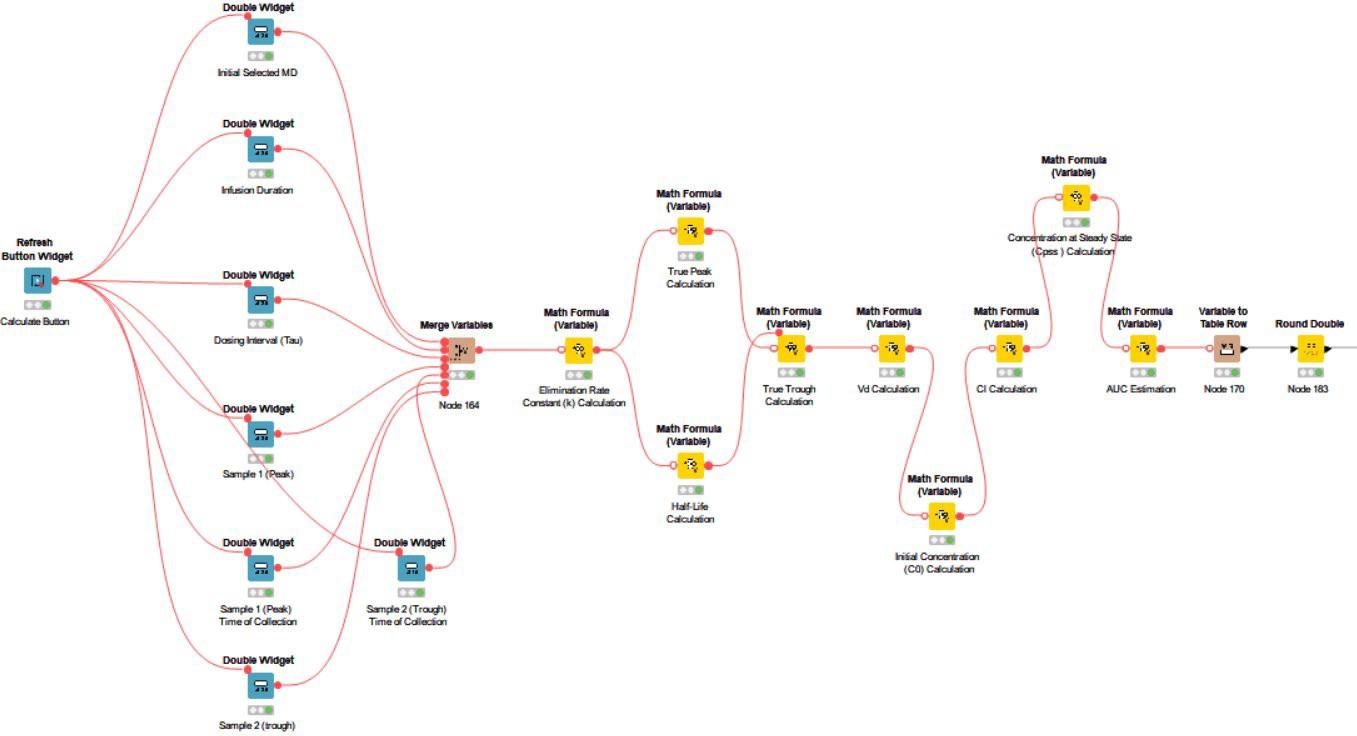
The workflow is split into three main routes. In Figure 2 you can see the pharmacokinetic calculations necessary for drugs administered intravenously (IV).
Here you can see how this part of the workflow looks when viewed via the interactive dashboard.
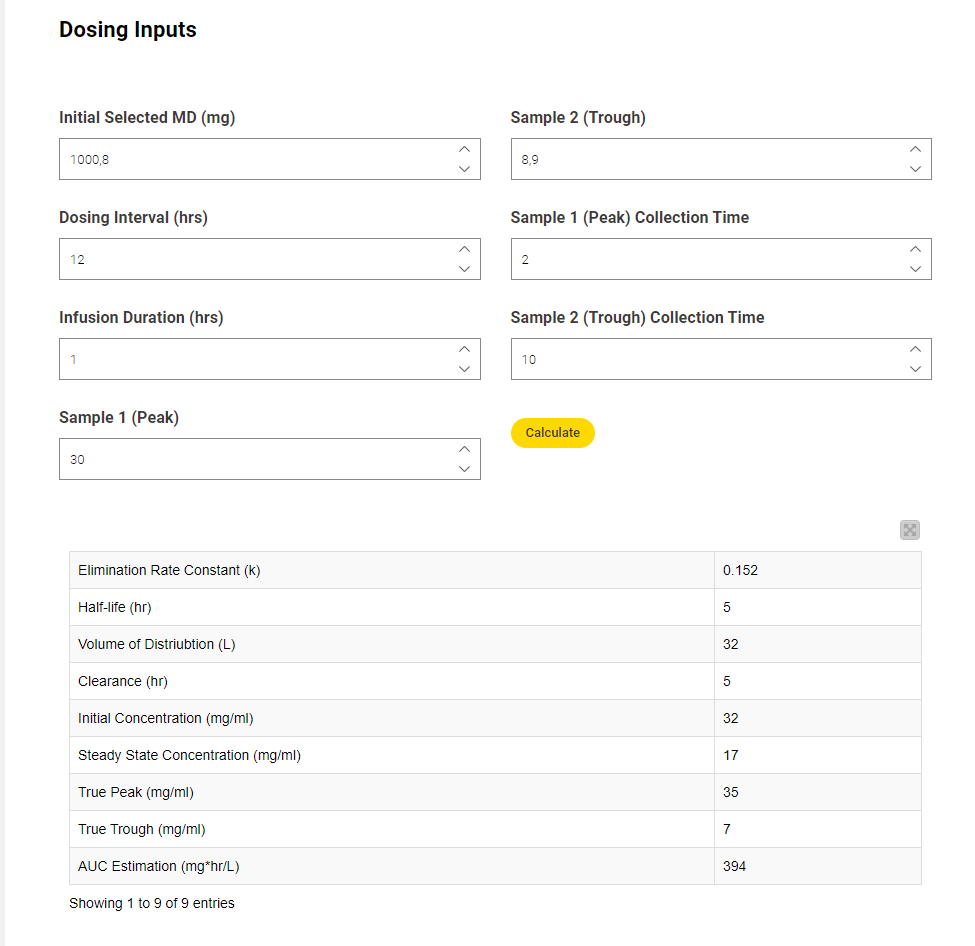
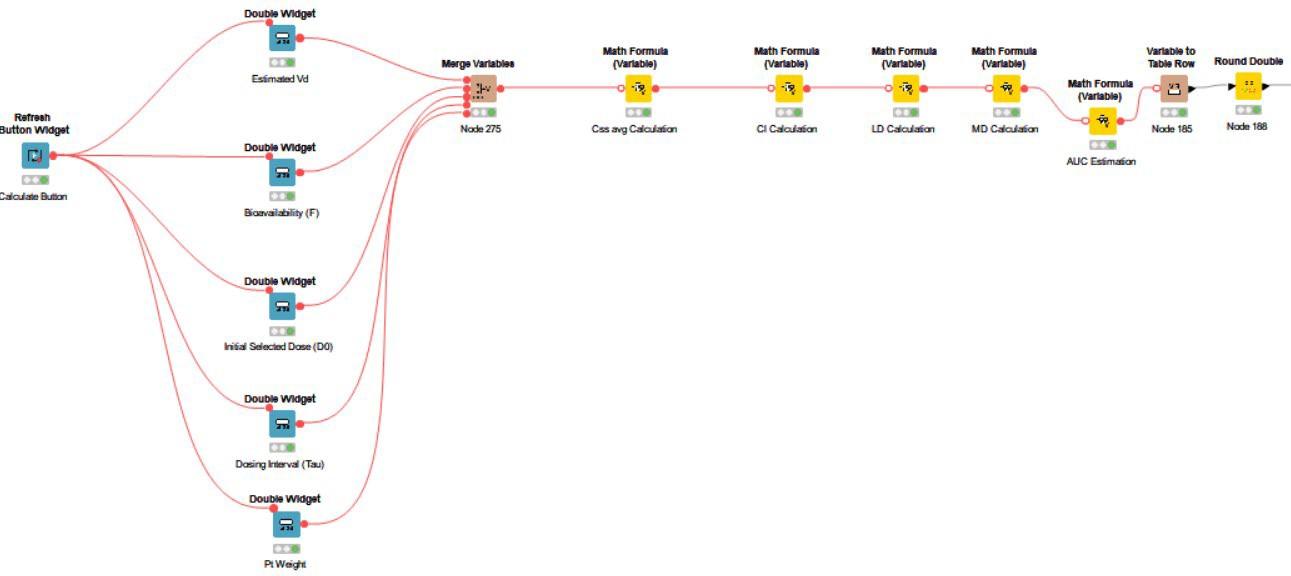
Figure 3 shows the calculations necessary for drugs administered orally (PO).
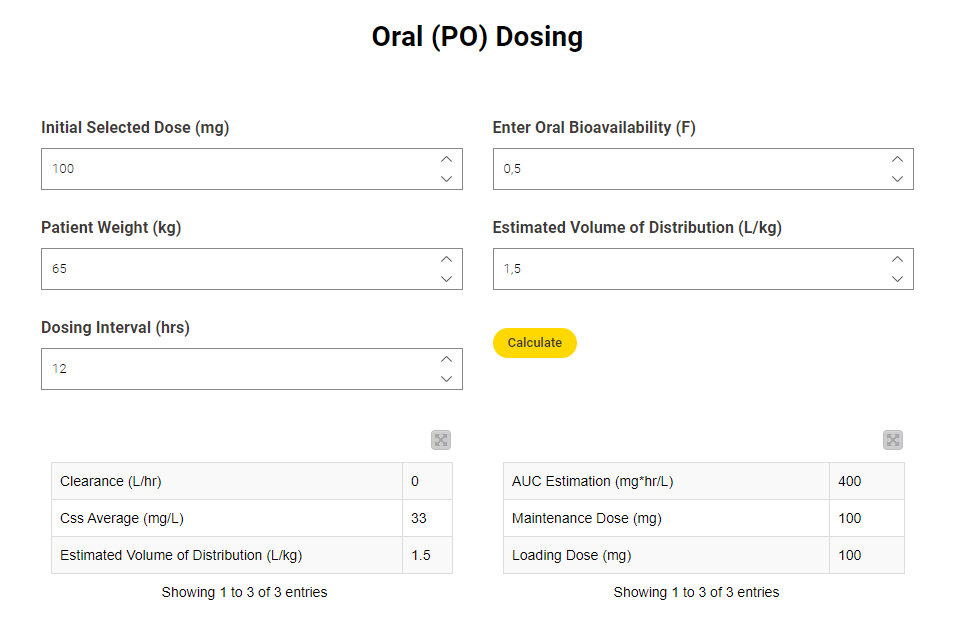
This workflow snippet underlies the part of the dashboard for entering and making the calculations based on orally administered drugs.
The calculation route, shown in Figure 4, takes care of a series of baseline calculations useful for dosing purposes.
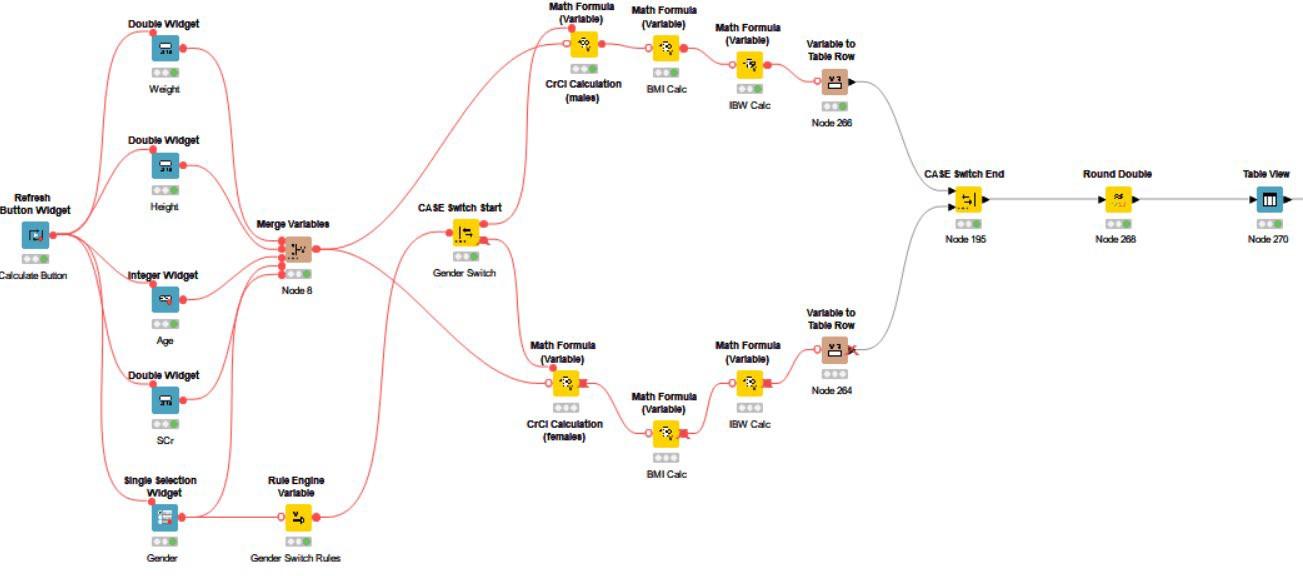
The blue widget nodes and table view nodes included in this workflow are the foundation of the interactive dashboard, allowing for patient baseline inputs and visualization of calculated data. The yellow math variable nodes allow for the embedding of necessary formulas for the pharmacokinetic calculations.
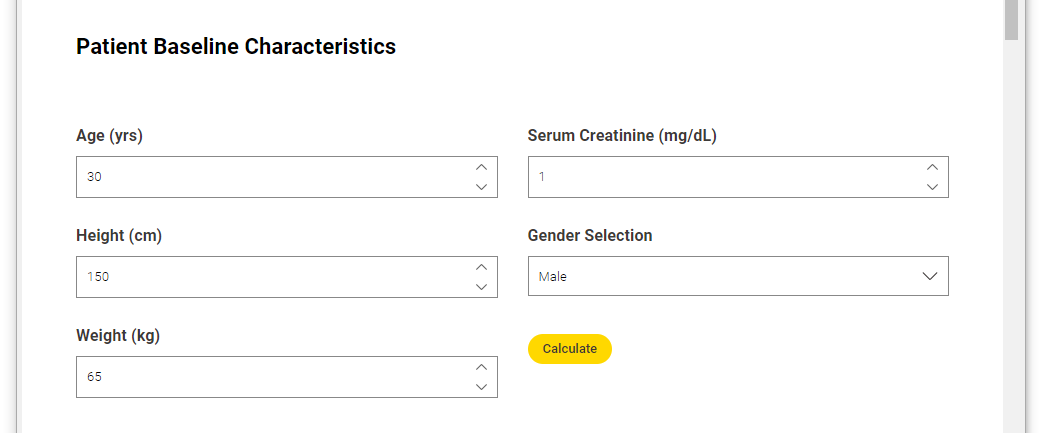
Here you can see how this part of the workflow looks when viewed via the interactive data app.
Digital Health Application Development
This project was part of an Advanced Pharmacy Practice Elective (APPE) rotation on Digital Health Application Development at Virginia Commonwealth University, School of Pharmacy. I used KNIME to build an automated solution. As a no-code/low-code data science tool, it gave me – as a healthcare specialist with no coding background – ease of learning and access to advanced analytics.
Get a walkthrough of the challenges of pharmacokinetics calculation and how I built this digital health application in the video below.
